


Molecular detection of minimal residual disease in blood
Molecular Detection of Minimal Residual Disease in Blood that Detects Earlier than Standard Techniques if Cancer is Still Present.
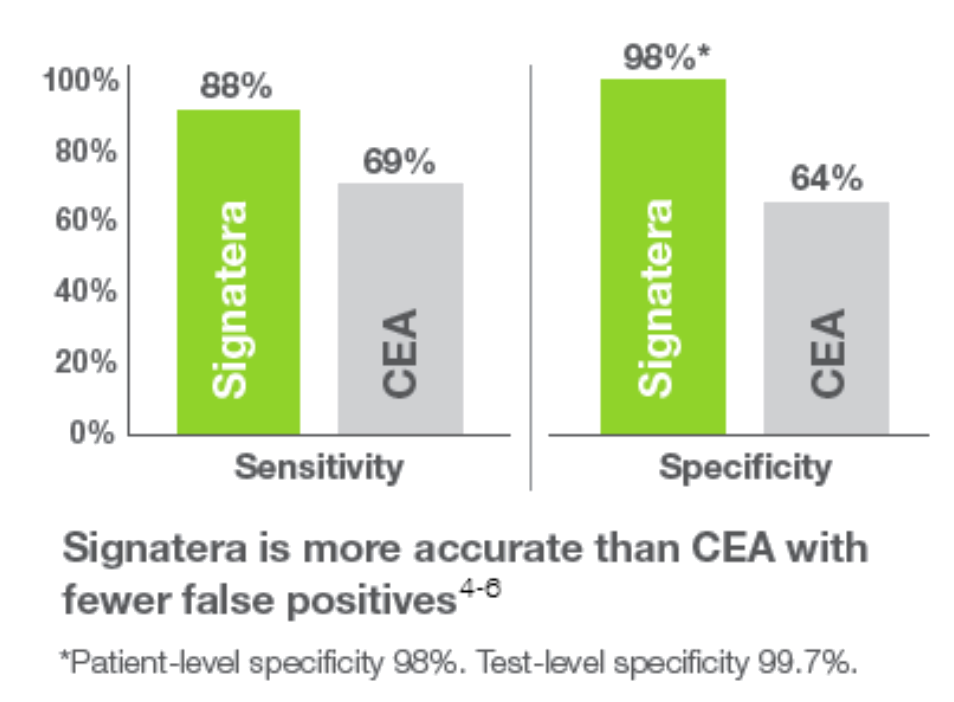
Signatera assay is an individualised, tumor-informed test that detects residual disease using circulating tumor DNA (ctDNA) in the blood of patients previously diagnosed with cancer.
Signatera detects residual disease in blood by testing a set of tumor mutations that are unique for each subject and can thus detect earlier than other traditional imaging methods if cancer is still present. This knowledge can help the treating physician to make treatment decisions more directly.
97% of patients with a positive Signatera result will relapse without additional treatment.
The test is initially performed in tissue and the detections are performed in blood where EDTA and Celll-Free DNA vials are required.
A positive test result indicates the presence of molecular residual disease (MRD). Serial sampling can be used to track whether disease burden is increasing or decreasing in response to treatment.
A negative test result indicates that molecular residual disease (MRD) was not detected. Repeat testing can be used to track whether disease burden remains undetectable or increases to detect relapse early

What is the turnaround time for my results?
Initial test takes 3-4 weeks and repeat testing has a turnaround time of 7-10 business days.
What type of sample is required for the test?
A tissue sample will need to be provided first and then EDTA and Cell-Free DNA vials with patient’s blood. Vials are provided by Genekor.
Is the test covered by any public/private insurance?
Fore cost coverage and related information, please contact the secretariat here.
How do I make the payment for the test?
Payment can be made by bank transfer or debit/credit card.
How I send the sample and receive the results?
Regarding the receipt of the sample, please contact the secretariat. The results are shared with your doctor and with you via email.
Our Customer Service Team is committed to answer your questions with regards to the services offered by Genekor. If you would like to order any of the tests that Genekor performs please contact us directly.
*To complete the test, you are required to complete and send the Consent form that you will find on the link below.
*For more information on scientific content please contact: scientific.support@genekor.com
*Download the promotional brochure here.